- Blog
- Icecream screen recorder activation code
- Hrd test
- Zombie frontier 3 hack unlimited cash gold download
- Midtown madness 1 mods
- Geometry dash on ti 84 plus ce
- Download radioboss full
- Answer bump of chicken lyrics
- Maithili malayalam actress hot photos
- Download any emulator bios apk
- What is sudarshan kriya yoga
- Pancha sinhala cartoon 2012
#HRD TEST HOW TO#
How to recognize this patient who do not have the BRCA mutation, but HRD.
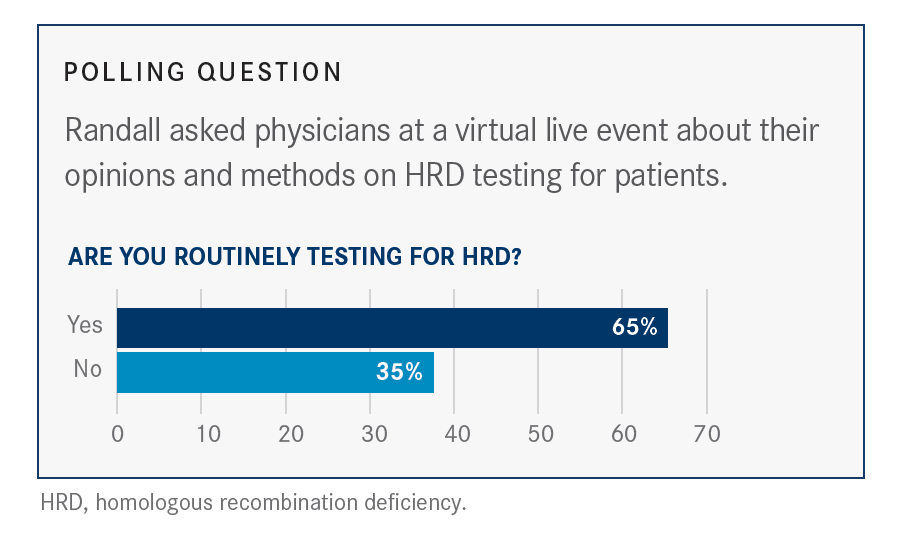
the patients who have an HRD-positive test, who has a normal reverse recombination repair deficiency, but have not a BRCA mutation. And this is new, why? Because until now, the population that benefits most from a PARP inhibitor was mainly those patients with a BRCA mutation, and there, it happens, a new subset of patients and new population. It is in line with the mechanism of action of a PARP inhibitor. And the remarkable finding is that it’s mainly those patients with HRD, tested by the Myriad tests, who benefit from the PARP inhibitor. But since the ESMO Congress, 3 randomized phase 3 trials have shown that the addition of a PARP inhibitor prolongs significantly the progression-free survival (PFS) of these patients who have high-grade ovarian cancer. PUJADE-LAURAINE: Until recently, the treatment of advanced ovarian cancer consisted of the combination of chemotherapy and debulking surgery-either upfront debulking surgery or interval debulking surgery. TARGETED ONCOLOGY: Prior to these 3 studies, what outcomes were observed with platinum-based chemotherapy and debulking surgery alone for the frontline treatment of patients with ovarian cancer? Pujade-Lauraine, head of the Medical Oncology Department at Hôpital Hôtel-Dieu in France, discussed PARP inhibitors and the benefits of various testing methods for HRD, in an interview with Targeted Oncology ahead of the SGO meeting. In a presentation during the Society of Gynecologic Oncology (SGO) 2021 Annual Meeting on Women’s Cancer, Pujade-Lauraine et al demonstrated data showing that these gene panels are not truly predictive of benefit from the combination of olaparib and bevacizumab in patients with ovarian cancer. However, according to Eric Pujade-Lauraine, MD, PhD, this method identifies fewer patients with high-grade ovarian cancer who have mutations in homologous recombination repair (HRR) pathway genes than the Myriad test. In addition to HRD testing with the Myriad assay, though, HRD testing with gene panels is another approach.

And the study demonstrated that patients with HRD positivity, regardless of BRCA mutation status, benefited most from the combination regimen.
#HRD TEST TRIAL#
Participants in the trial were tested for tumor HRD status using the Myriad m圜hoice HRD Plus assay, which was later approved by the FDA as a companion diagnostic for olaparib. This indication was granted based off of findings from the phase 3 PAOLA-1 trial (NCT02477644), which aimed to see if patients received any benefit from olaparib added to bevacizumab, regardless of BRCA mutation status.
The PARP inhibitor olaparib was also approved in combination with the angiogenesis inhibitor bevacizumab (Avastin) as a first-line maintenance therapy in adult patients with ovarian cancer who are in complete or partial response to first-line platinum-based chemotherapy and whose cancer is associated with homologous recombination deficiency (HRD), as defined by either a deleterious or suspected deleterious BRCA mutation, and/or genomic instability. PARP inhibitors such as olaparib (Lynparza), niraparib (Zejula), and rucaparib (Rubraca) have all been approved by the FDA and the European Medicines Agency as first-line maintenance therapies for patients with advanced ovarian cancer. For patients with homologous recombination repair deficiency (HRD), PARP inhibitors can be an effective treatment for patients with ovarian cancer.
- Blog
- Icecream screen recorder activation code
- Hrd test
- Zombie frontier 3 hack unlimited cash gold download
- Midtown madness 1 mods
- Geometry dash on ti 84 plus ce
- Download radioboss full
- Answer bump of chicken lyrics
- Maithili malayalam actress hot photos
- Download any emulator bios apk
- What is sudarshan kriya yoga
- Pancha sinhala cartoon 2012
